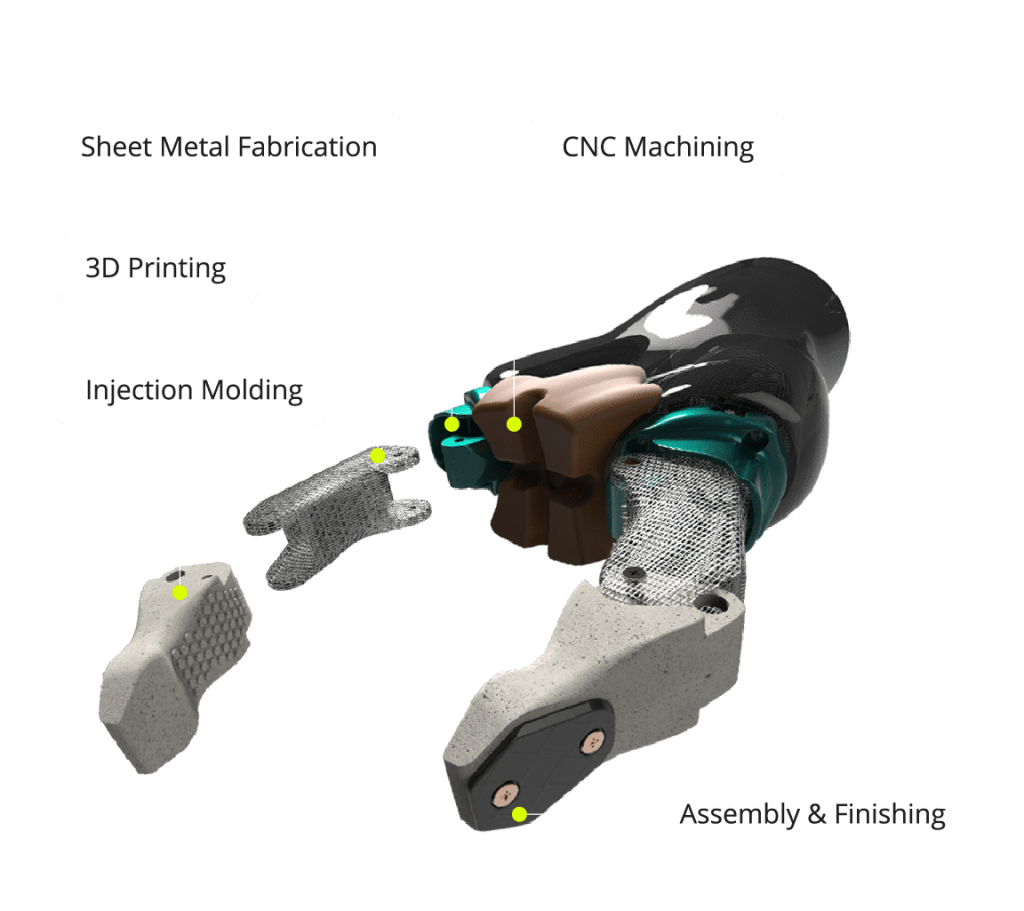
Medical
Trusted by Industry Leaders




Move Faster with Proven Medical Expertise
Medical device teams operate under strict quality requirements and documentation expectations. Risks are high and tolerance for variation is low. Challenges often include maintaining process consistency, supporting traceability, meeting cleanliness requirements and ensuring suppliers can align to regulated workflows.
We deliver comprehensive manufacturing support built on decades of medical expertise, stringent quality systems and tightly controlled processes that help medical OEMs achieve traceability, regulatory alignment, and production confidence.
-
ISO-certified quality systems – ISO 13485 certification and rigorous documentation ensure consistent, compliant medical manufacturing.
-
End-to-end support – From prototyping through production, we maintain process consistency across every stage.
-
Precision at scale – Advanced technologies and materials expertise enable tight-tolerance, traceable medical components.
Our Expertise – By the Numbers
ISO 13485:2016
110,000+
1,175+
25+
How Do Medical Companies Use Fathom?

Built for Regulated Environments
We operate under robust quality systems, with disciplined documentation, validation and process controls to support regulatory and compliance requirements.

Precision, Traceability & Processing Expertise
Advanced manufacturing technologies, material expertise and traceability controls enable us to produce tight-tolerance medical components while supporting cleanliness and quality expectations.

Consistency from Prototype to Production
Integrated engineering and manufacturing capabilities help medical teams maintain process consistency, manage risk and reduce variation as programs scale.
Fathom Capabilities for the Medical Industry

Medical-Grade Injection Molding
Fathom provides custom injection molding for medical components that require tight tolerances, repeatability and consistent material performance. This process supports a wide range of medical applications, from device housings to disposable components.

Precision CNC Machining
CNC machining enables the production of highly accurate medical prototypes and low-volume production parts from engineering plastics and metals. This capability allows medical teams to validate designs, test functionality and maintain dimensional accuracy before and during production.
Rapid Prototyping & Bridge Manufacturing
Fathom supports fast prototyping and bridge manufacturing to help medical product teams move quickly while managing risk. Flexible volumes and quick turnaround help teams meet development timelines without compromising quality or compliance expectations.

Rapid Quoting Plus Fast-Turn Manufacturing
When you’re trying to keep a new product launch on track, you need an agile manufacturing partner that can move as fast as you do. Fathom can help you keep your time-critical programs moving fast with 30-second quoting and parts shipped in as little as 15 days.
Numerous Material & Finishing Options
From post-machining and powder coating to assembly and packaging, we can customize secondary processes to meet your requirements. Our extensive finishing capabilities can save you time, money and factory space and help you bring your medical products to market faster.
A Long Track Record of Success
Medical plastic injection molding requires a level of expertise that only comes with decades of experience. Fathom has been providing low-cost injection molding and other manufacturing services to medical device manufacturers for over 20 years.
Common Medical Applications
Fathom supports medical programs with strong process discipline, defined quality checks and documentation support aligned to what regulated teams typically need.
We focus on repeatable manufacturing and clear communication so you can move confidently through development and into production. We give you the best of both worlds: low-cost overseas production plus U.S. engineering and project management.
-
Device housings and enclosures
-
Handheld components and grips
-
Cartridges, caps, and covers
-
Disposable component parts
-
Fit-critical assemblies and subcomponents
-
Lab and diagnostic device components
-
Service panels and access components
Why Medical Device Manufacturers Trust Fathom
Fathom supports medical device manufacturers with ISO-certified processes and a comprehensive manufacturing platform built for quality, speed and compliance.
By combining engineering, prototyping and production expertise, Fathom helps medical device teams move efficiently from development to production while reducing risk and meeting demanding medical requirements.
-
End-to-End Manufacturing Capabilities
From rapid prototyping to high-volume production of precision parts, Fathom supports the full product lifecycle within one trusted manufacturing partner.
-
Fast Turnaround for Time-Critical Programs
Streamlined quoting, in-house tooling and global manufacturing options enable quick lead times ideal for tight development or production schedules.
-
Expertise in Medical-Grade Materials
Fathom works with a wide range of FDA-compliant and medical-grade plastics, helping manufacturers meet performance, safety and regulatory requirements.
-
DFM Support
Engineering teams provide early design for manufacturability (DFM) feedback to reduce risk, control costs and optimize parts for reliable, repeatable medical production.
Launch Your Injection Molding Project with Confidence
Partner with Experts Who Get It Right the First Time
Every project is reviewed by a seasoned manufacturing engineer to optimize part design, material selection, and tooling strategy — ensuring smarter decisions before steel is cut and production begins.